Graphene has very high surface area per unit weight
Possesses high electron mobility
Remarkable bactericidal activity
200x stronger than steel
Retains elasticity and flexibility despite strength
Graphene’s remarkable properties allow it to be applied to a broad range of raw materials
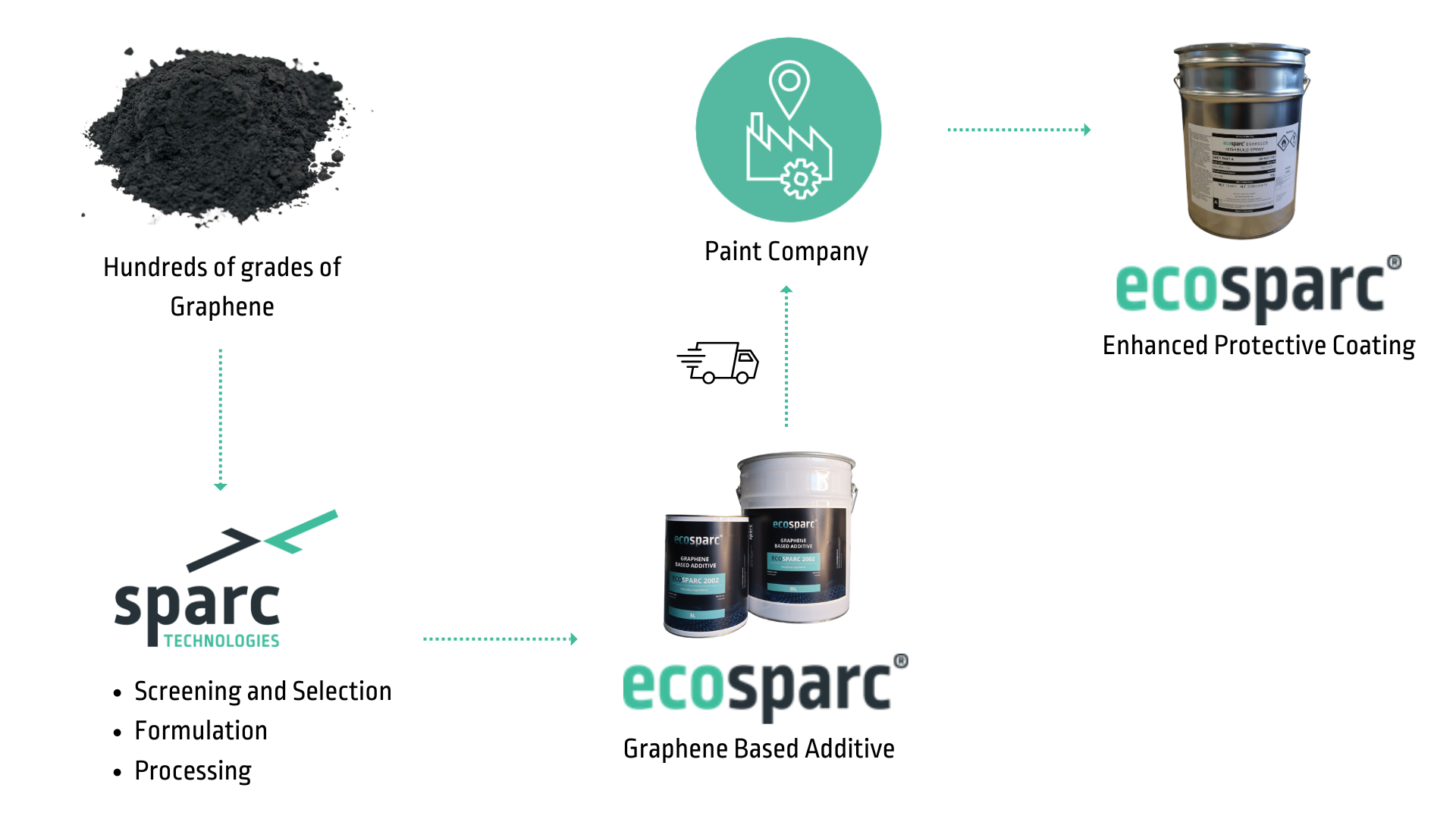
Not all graphene is the same - graphene must be “functionalised” for specific purposes
Good affinity with non-polar systems

How Graphene is made
There is a variety of ways of making graphene, utilising different feedstocks.
Graphite – Various methods of graphite exfoliation isolate single to few layers of graphene. The properties of graphene obtained from this method is variable both in its physical form and associated applications.
Chemical Vapor Deposition (CVD) – Utilised for graphene manufacture of high purity, where performance requirements are high.
Biomass – Graphene and graphene like materials can be sourced from a variety of biomass sources via pyrolysis.
You don't need much of it!
Positive impacts of graphene can be realised with addition rates as low as 0.1% by weight.
It's more conductive than graphite
Graphene is much more conductive than graphite due to the availability of electrons to carry charge.
It's super strong!
Addition of graphene increases strength, flexibility and elasticity.
It's a superconductor
Addition of graphene to something increases heat and electrical conduction.
Fun Fact:
The surface area from one gram of graphene is enough to cover roughly 3 basketball courts.
Sparc’s Unique Graphene Position